A Phase of Its Own
Plutonium’s unusual behavior forced early Los Alamos scientists to rethink metallurgy, laying the foundation for plutonium science as it exists today.
- Eleanor Hutterer, Editor

The small metal ball was freshly cast and still hot—100 grams of a brand-new, human-created metal that, on paper, should behave like uranium. The technicians who made it set it aside to cool. By the next day it had warped, cracked, and split in two. No one had touched it, yet the plutonium had changed. At Los Alamos, in early January of 1945, this wasn’t just an inconvenience, it was a crisis.
For nearly two years, scientists at Los Alamos had been working feverishly on the Lab’s mission of producing a plutonium-based atomic bomb. But that wartime race would be won or lost on a single hurdle: the mastery of plutonium.
Virtually nonexistent in nature, plutonium was first synthesized in late 1940 and was the Manhattan Project’s first-choice material for an atomic bomb. In mid-1944 the original gun-style weapon design was scrapped because it wouldn’t work with plutonium due to high spontaneous fission, and the scope shifted to a more complex implosion-style design. But as Lab scientists desperately tried to work with it, plutonium’s weird behavior, defying basic metallurgy and common sense, was slowing progress down.
In the spring of 1945, key pieces of the puzzle finally started coming together, and mere months later, the Lab delivered the first atomic weapon, an implosion-style bomb with plutonium at its heart. The histories of Los Alamos and plutonium have been intertwined ever since—one would truly not exist without the other. In understanding and harnessing this new human-made element whose behavior was so bizarre, the Lab became the world’s center for plutonium science. Nearly every foundational discovery in plutonium metallurgy and materials science over the past eight decades was either made at the Lab or motivated by its science.
Discovery
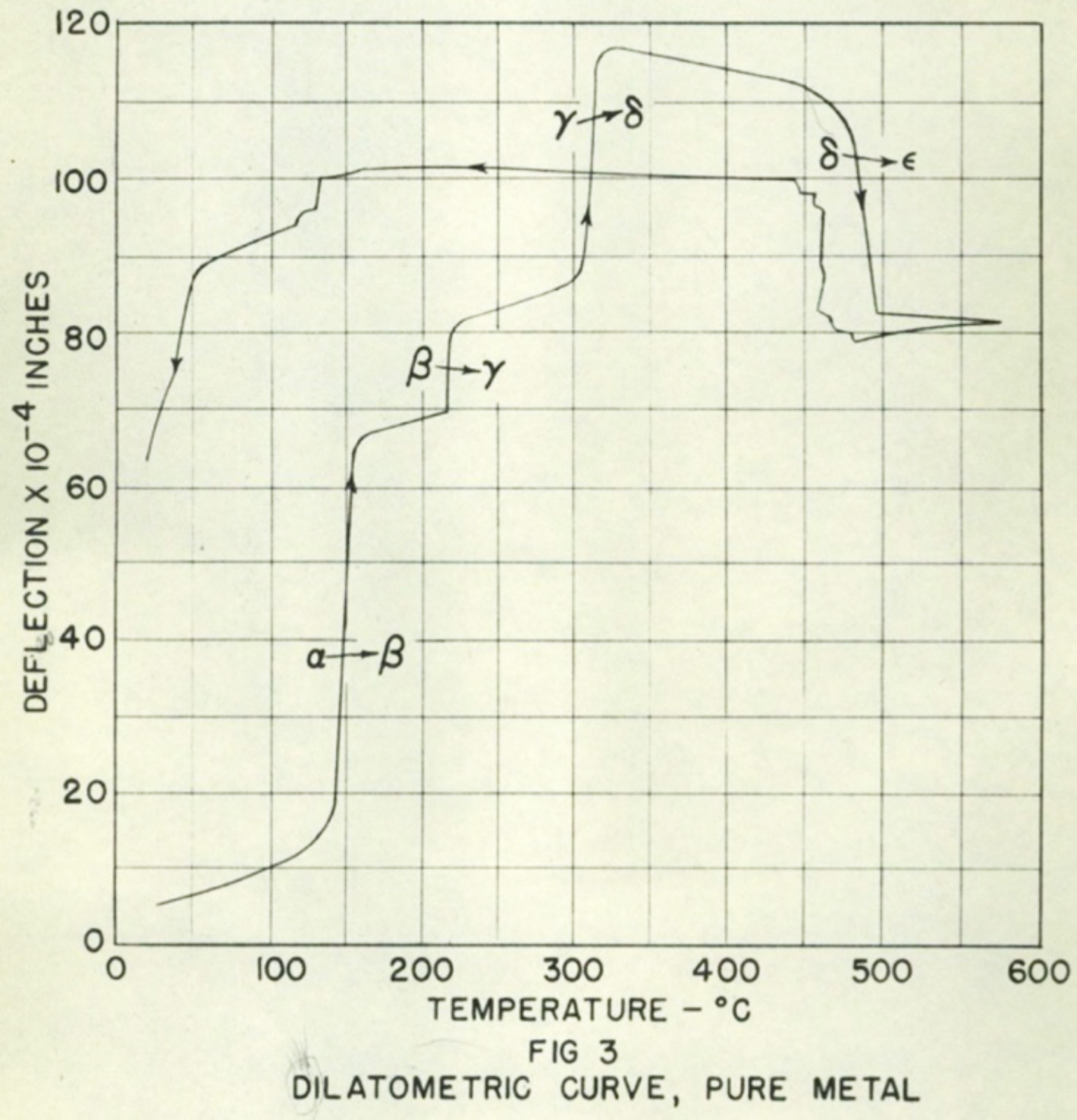
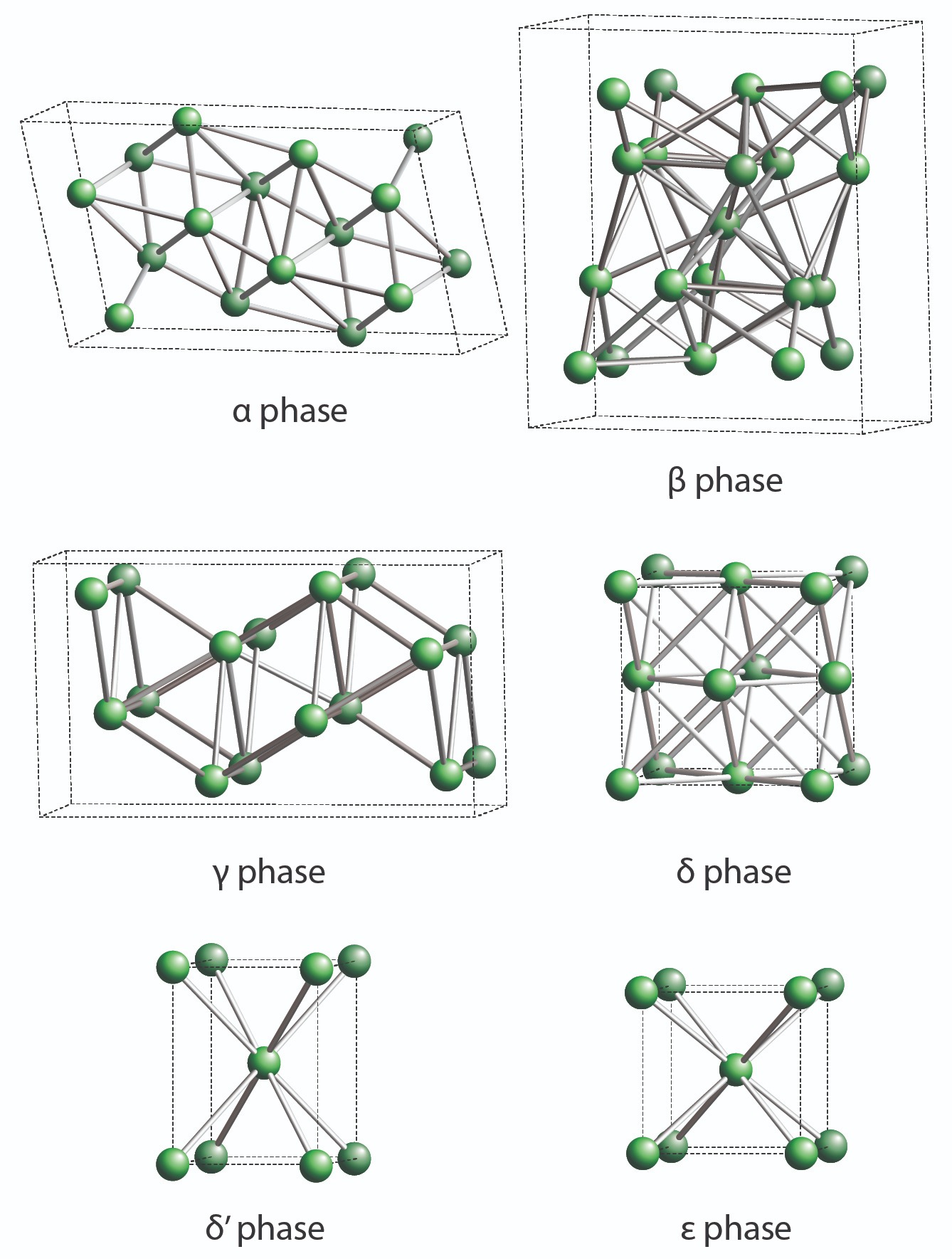
Plutonium has been described as a physicist’s dream and an engineer’s nightmare because of its superb nuclear properties and confounding physical properties. Compared to uranium, plutonium is more efficient at fission, meaning far less material is needed to achieve a nuclear chain reaction. But it has six different phases, or crystalline forms, that vary in density by up to 25 percent, and modest changes in temperature or pressure can provoke it to go from being as malleable as aluminum to as brittle as glass. Maddeningly, its density can decrease as temperature increases, the opposite of how most elemental metals behave. These dramatic changes in density as plutonium transforms from one phase to another is why that first 100-gram ball cracked—its volume collapsed suddenly as it cooled into a denser phase.
Plutonium has been described as a physicist’s dream and an engineer’s nightmare because of its superb nuclear properties and confounding physical properties.
Because plutonium’s nuclear properties are so ideal, Manhattan Project scientists needed to find a way to make it cooperate. The implosion design demanded metal parts that were dimensionally stable with precisely known densities, but as time passed and temperature changed, the plutonium kept quietly rearranging its internal structure. One strategy was to alloy it, to mix in another element that could stabilize it in its most workable phase, the delta phase. And so began The Plutonium Alloy Survey Program, a rapid, systematic search for phase stabilizers.
“They threw the kitchen sink at it,” says Lab Fellow and retired engineer Joe Martz. “They would combine various elements, up to five at a time, in a little plutonium casting, then look for a density reduction that would indicate the delta phase was stabilized.”
Initially they tested about thirty elements. Two of them, silicon and aluminum, showed promising density reduction, so those were pursued further. Aluminum emerged as the frontrunner, creating a beautifully stabilized delta phase plutonium alloy that was easy to machine and didn’t crack when it cooled.
Then in March of 1945, Los Alamos luminary and future Noble Prize winner Hans Bethe wrote a paper on the limits of plutonium as a fissile material, from a nuclear physics standpoint. It showed that the neutron background it could tolerate was below what an aluminum alloy would create, meaning to use aluminum would risk pre-detonation or outright fizzle. The best path forward was suddenly and decisively blocked.
The design demanded metal parts that were dimensionally stable, but the plutonium kept quietly rearranging its internal structure.
“Someone—maybe several scientists discussing—suggested they look at gallium, the element immediately below aluminum on the periodic table,” says Martz. “Because of the way the table is organized, gallium shares many chemical and physical characteristics with aluminum.”
It was an inspired choice, and within weeks they showed that gallium would make an even better plutonium alloy than aluminum. It was stable, comparatively easy to work with, and crucially, didn’t create a large neutron background. Gallium was so good that they ended The Plutonium Alloy Survey Program in April, finalized the implosion-style design in May, and delivered the first plutonium driven atomic bomb in July.
Details
In less than two years the Lab had developed an entirely new metallurgy discipline to take plutonium from barely visible samples to kilogram-scale parts, and that groundwork laid the foundation for all later plutonium science and even the first plutonium-fueled reactor. Post war there were still more questions than answers about the temperamental metal, so Los Alamos scientists moved into more extensive examination.
Lab metallurgists knew that the alpha phase, the form of unalloyed room-temperature plutonium, was dense and brittle but they couldn’t explain why. By 1946, innovative x-ray crystallographers had solved the crystal structures for several phases, revealing that in alpha phase, plutonium atoms are locked into a complicated monoclinic structure that doesn’t tolerate stress, making it brittle and crack-prone—more like a mineral than an ordinary malleable metal. Delta phase, on the other hand, has a simple cubic crystal structure more typical of most metals.
Also in 1946, the Lab built the Los Alamos Plutonium Fast Reactor, the world’s first plutonium-based nuclear reactor. It was nicknamed Clementine after the Gold Rush folk song “Oh My Darling, Clementine,” because it was built in a canyon near a cavern and its operators called themselves “49ers”, in a wink at plutonium’s wartime code name, “49.” Built to use metallic plutonium fuel, Clementine was a research tool for studying neutron cross sections, spectra, and high-energy effects. Clementine was designed in the immediate shadow of two fatalities caused by hands-on criticality experiments—Harry Daghlian in 1945 and Louis Slotin in 1946. Because of those tragedies, the reactor incorporated remote-handling safety systems, signaling an important shift in how the Lab approached dangerous work. Over the six years that it was operational, Clementine taught plutonium scientists an enormous amount about neutrons, plutonium, and reactors. Even when it failed—first in 1950 and terminally in 1952—it yielded valuable insights. The reactor was decommissioned when its plutonium metal fuel rods were found to have ruptured due to structural instability and irradiation damage.
After Clementine came the Los Alamos Molten Plutonium Reactor Experiments (LAMPRE I and II), an effort to develop a reactor that used molten plutonium, rather than solid, as its fuel. The idea was this: Clementine’s solid fuel rods failed as a result of irradiation damage and crystal lattice instability; molten metal doesn’t have a crystal lattice so doesn’t accrue irradiation damage; therefore, molten plutonium wouldn’t suffer the same effects. Theoretically, a molten plutonium reactor could run for a very long time. There was a large international effort in the 1960s around this idea, but it eventually stalled due to other material compatibility challenges and operational complexity.
Modern nuclear reactor programs still rely on the foundational data gathered from Clementine and the LAMPRE program. Nuclear reactor research moved away from using pure plutonium as fuel, developing instead, uranium and uranium-plutonium alloys, oxides, nitrides, carbides, or molten salts as safer, longer lasting fuels.
In less than two years the Lab developed an entirely new metallurgy discipline to take plutonium from barely visible samples to kilogram-scale parts.
Shock physics and studies of how plutonium behaves under extreme pressure also ramped up steadily over the years. The 1970s and 80s saw innovative advances in measurement science, especially for measuring critical dynamic properties like strength and compressibility. New techniques were developed, like explosives-driven shock experiments and radiography methods that were first developed on plutonium analogs, then later used for plutonium under tight control.
Dynamics
Over the last 35 years or so, focus has been on refining the measurements made during the early years and understanding plutonium aging. Los Alamos continues to lead the way in this innovative science, which is among the most vital activities supporting nuclear deterrence.
Plutonium is still alloyed with gallium to make it workable, and it’s important to know how those atoms are arranged and whether, as with Clementine, irradiation damage is building up. In the 1990s and 2000s, accelerated aging methods—like spiking plutonium-239 with a more radioactive plutonium isotope to achieve faster irradiation damage—were developed. Dynamic deformation measurements and subcritical experiments in Nevada were also key in the study of plutonium aging. Enough new data was generated during this time to warrant a massive update to The Plutonium Handbook, the authoritative technical guide to plutonium science, expanding it from its original two volumes in 1967, to seven volumes in 2019.
A boon to plutonium science came in 2012, when researchers at Los Alamos achieved the first successful nuclear magnetic resonance measurements of plutonium-239. Nuclear magnetic resonance detects the specific radiofrequency that atomic nuclei absorb in a strong magnetic field. Observing this signal in plutonium opened the door to powerful atom-scale probes of electronic structure and local atomic disorder—both central to understanding how self-irradiation affects plutonium.
Importantly, Los Alamos played a vital scientific and technical advisory role in the 1992 closure of the Rocky Flats Plant, near Denver, where plutonium for weapons had been fabricated since 1952. The plant, which was government owned but privately operated, was closed due to environmental concerns. Los Alamos scientists were able to show that the chemical form of plutonium contaminating the site was not soluble in water, and therefore not migrating into subsurface groundwater; however, it was able to be transported by surface soils through wind and surface water erosion. This meant removing topsoil would be an effective cleanup strategy, which indeed it was. The site is now a wildlife refuge.
With Rocky Flats closed, plutonium manufacturing has now returned to Los Alamos, where it all started. “We need to keep science and engineering and production tightly coupled,” says chemist and Lab Fellow Dave Clark, who was on the Rocky Flats clean up advisory team. “When manufacturing is isolated from science, failures happen and scientists get brought back in to diagnose.”
Plutonium remains vital to national security and irresistible to science. “Beyond its nuclear utility, the science is fascinating,” Clark says. “Plutonium’s exotic electronic structure gives rise to highly correlated 5f electron phenomena that drive condensed-matter questions and a lot of theory effort.” Put simply, plutonium is still one of the most challenging and scientifically productive elements on the periodic table.
Los Alamos was built around the need to domesticate plutonium. Eight decades later, the metal is still strange, still stubborn—and still at the center of the Lab’s mission.
People Also Ask:
- Is plutonium manmade? Effectively, yes. Trace amounts of plutonium isotope 244 have been reported in natural uranium ores and on the ocean floor, but not in extractable or usable quantities. Plutonium isotope 239 for nuclear weapons and nuclear power applications is made by bombarding uranium isotope 238 with neutrons. When a uranium-238 atom (atomic number 92) absorbs a neutron, it undergoes beta decay and becomes neptunium-239 (atomic number 93), which, upon absorbing another neutron, beta decays into plutonium-239 (atomic number 94).
- What is plutonium used for? Different isotopes of plutonium are useful for different applications. Plutonium isotope 239 is most famously used in nuclear weapons and as fuel for some nuclear reactors. Plutonium isotope 238 is used to power NASA space probes and rovers and was used during the 1970s to power long-lived cardiac pacemakers, though this application was phased out in the late 1980s and replaced with lithium batteries.








