Proton Power for Public Health
How Los Alamos turned nuclear science into a medical marvel
- Eleanor Hutterer, Editor

In the 1940s, as Los Alamos scientists raced to unlock and harness the physics of fission, a parallel scientific revolution was taking shape, one that would take nuclear science from splitting atoms to saving lives: biomedical isotopes.
The science of isotopes was fundamental to the mission at Los Alamos from day one. Some isotopes fission readily, like the uranium used in nuclear reactors (U-235), while others don’t, like the uranium found in nature (U-238). The difference boils down to neutrons. Isotopes of a given element have the same number of protons (92 for uranium), but different numbers of neutrons (143 for U-235 and 146 for U-238), meaning they behave the same chemically but differ in mass or radiation emissions.
These differences mean both stable and unstable isotopes can be used as labels. Stable isotope mass measurements can be compared to identify the ratio of particular isotopes that is present. Unstable isotopes emit radiation that can be detected. These characteristics make isotopes powerful, nondisruptive tracers and probes for living systems. As Manhattan Project scientists formed a deep practical understanding of isotope separation, enrichment, and measurement methods, visionary researchers saw vast potential in biomedicine. Through the 1940s and 1950s, Lab scientists turned that understanding from wartime nuclear physics toward broad, exploratory science.
Stable isotopes
Los Alamos was one of the world’s first producers of enriched stable isotopes, which are useful as nonradioactive molecular tracers. When measured as ratios of heavy to light, like carbon-13 to carbon-12, stable isotopes can reveal information about metabolic or pathologic processes. They can also be used to selectively strengthen otherwise weak atomic signals, allowing new types of nuclear measurements.
Carbon-13, for example, was essential to the development of nuclear magnetic resonance (NMR) spectroscopy, which enables today’s magnetic resonance imaging (MRI) and a slew of other applications in which precise, non-destructive chemical and structural measurement is needed. Carbon-12 is the dominant form of carbon in nature, but because it has no magnetic moment, it’s invisible to NMR, which uses magnetic field maneuvers to detect certain nuclei. So, to make biochemicals measurable by NMR, some of their carbon-12 atoms have to be replaced with carbon-13, which has an unpaired nuclear particle, giving its nucleus a magnetic moment and making it visible to NMR. Throughout the 1950s, Los Alamos advanced both the science and the supply chain that took NMR from a physics curiosity to a cornerstone technique in chemistry, biology, and medicine. Other key spectroscopic, or molecular-interaction, techniques developed during this time that relied on Los Alamos–made stable isotopes include Raman spectroscopy, electron spin resonance, and surface plasmon resonance.
Throughout the 1950s, Los Alamos advanced both the science and the supply chain that took NMR from a physics curiosity to a cornerstone technique in chemistry, biology, and medicine.
During the 1970s and 1980s, Los Alamos pioneered the large-scale isolation of stable isotopes using enormous distillation columns, the largest extending down nearly 700 feet into the ground. The columns were so tall because they used evaporation and condensation to physically separate isotopes by mass, but because the mass difference was so slight—just one neutron—it took many cycles of inching the lighter isotopes toward the top and the heavier isotopes toward the bottom to get adequately pure populations. For more than 20 years, the Lab was home to the National Institutes of Health’s Stable Isotope Research Resource, which supported frontier research in basic biology and biomedicine. While production of stable isotopes at Los Alamos has dwindled, having been successfully transitioned to industry, production of unstable isotopes, or radioisotopes, continues in force today and powers vital medical imaging and treatment on a global scale.
Unstable isotopes
Radioisotopes decay at predictable rates, becoming other isotopes or even other elements, by emitting different types of radiation. It’s the radiation that enables medical imaging and therapy. Rubidium-82 (Rb-82), for example, is used in cardiac PET (positron emission tomography) imaging and allows real-time visualization of blood flow and heart function. But Rb-82 is short-lived, lasting just over a minute. So, the trick is to start with strontium-82 (Sr-82), also a radioisotope but one that lasts weeks rather than minutes. The pathway goes like this: unstable Sr-82 converts a proton into a neutron and emits a neutrino, thus becoming Rb-82; Rb-82 is also unstable and converts another proton into a neutron, emits another neutrino and a positron, and stops decaying as a stable isotope of krypton. The emitted positron is what enables medical imaging, but because Rb-82 decays so quickly, it has to be produced onsite from a Sr-82 source as needed for PET imaging. The ability to produce, purify, and distribute large quantities of Sr-82 fundamentally changed how coronary artery disease is diagnosed and managed, and Los Alamos made it happen.
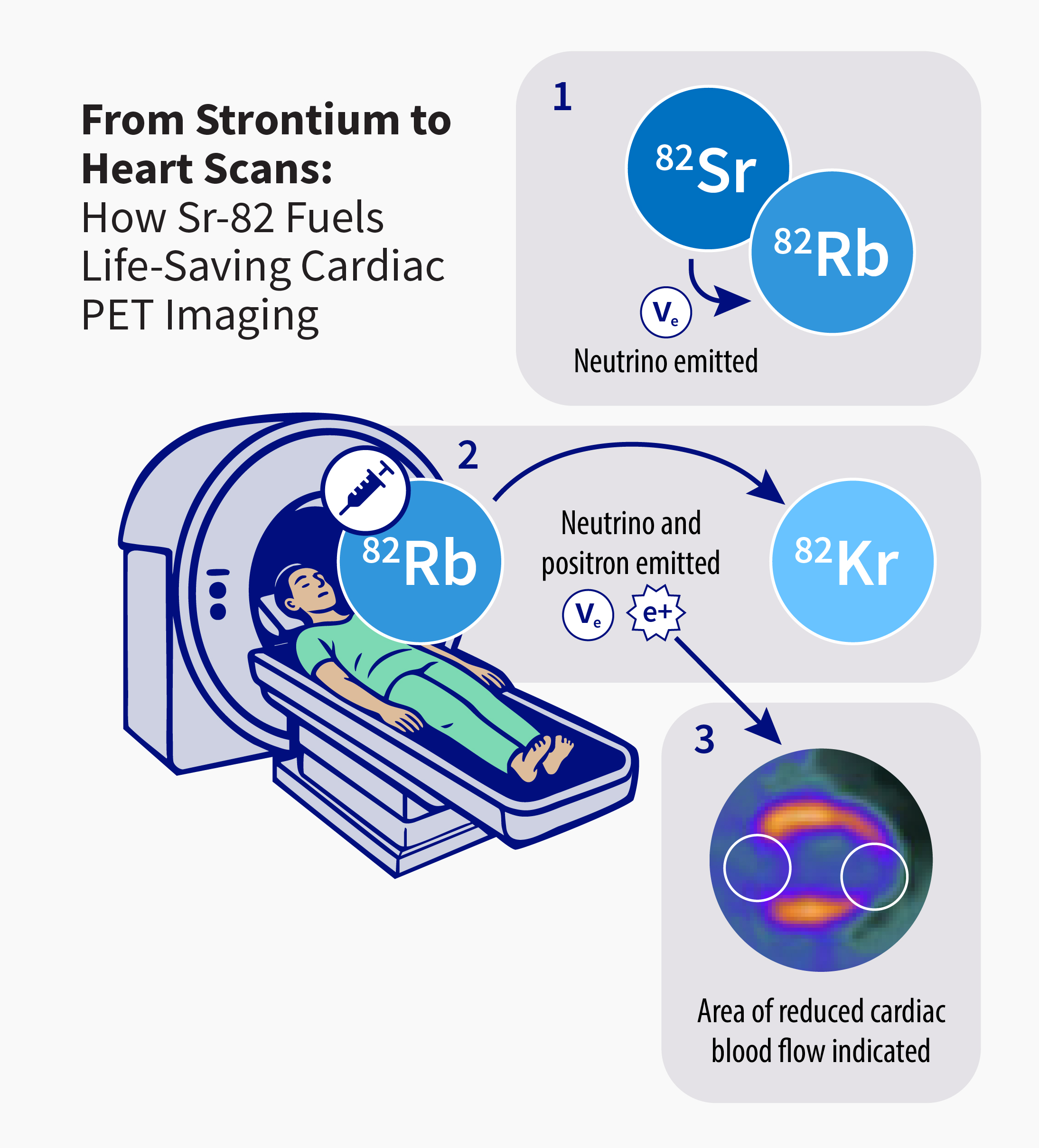
(1) A Sr-82 nucleus is proton rich and unstable, so it undergoes a process called electron capture: an inner atomic electron is absorbed, causing a proton to become a neutron, thereby changing the nucleus from Sr-82 (38 protons and 44 neutrons) to Rb-82 (37 protons and 45 neutrons). The process also results in the emission of an electron neutrino.
(2) The Rb-82 is injected into a patient who is inside a PET scanner. Rb-82 is still not stable, so again an electron is absorbed, a proton becomes a neutron, and a neutrino comes out. But this time, a positron is emitted along with the neutrino, and the nucleus, now krypton-82 (Kr-82; 36 protons, 46 neutrons), is finally stable.
(3) Positron emission is the key to cardiac imaging. After injection, Rb-82 is quickly absorbed by the heart muscle in proportion to blood flow. As Rb-82 decays into Kr-82, each emitted positron travels a couple millimeters then annihilates with an electron to generate two gamma photons. These photons are detected by the PET scanner and used to construct high-resolution images showing any areas of reduced blood flow, a sign of coronary artery disease. This β⁺ decay process, as it’s known, and the short half-life of Rb-82, powers safe, high-speed, high-resolution modern cardiac imaging.
Before the 1970s, the Lab had a cyclotron accelerator to produce protons for physics research. Isotopes were made at the end of the beamline where the protons were dumped afterwords. But in 1972, the substantial radiochemistry capability that had amassed at Los Alamos was formalized into the Medical Radioisotope Program and backed by a proton accelerator within the brand new Los Alamos Meson Physics Facility. Designed to study subatomic particles, the same accelerator could fire high-energy protons into metal targets to force a redistribution of neutrons among atoms and create more isotopes more easily.
Sr-82 was one of the first isotopes the program produced, but making it wasn’t the only challenge—equally difficult was extracting it from the cocktail of radioactive fragments that came off the beamline. Lab scientists had to invent new purification chemistry to separate the micrograms of useful Sr-82 from the kilograms of irradiated target material. In the late 1970s, Los Alamos became the first site to routinely produce Sr-82 for medical imaging.
The Medical Radioisotope Program grew through the 1980s, and in 1988 the Department of Energy’s Office of Nuclear Energy launched the Isotope Production and Distribution Program (now the DOE Isotope Program) to consolidate isotope production and make it self-supporting through commercial sales. The program grew into a world-class and reliable supplier of both Sr-82 for cardiac imaging and germanium-68 as a calibration standard for PET imaging, as well as a broad suite of more unusual isotopes including copper-67 for cancer treatment.
“The Lab’s accelerator-based isotope supply chain supports research institutions and commercial partners across the U.S. and lends resilience against foreign dependence or global shortages.”
By the late 1990s, demand continued to grow and the program needed its own purpose-built proton beam to produce more isotopes more efficiently and at greater purity. So, in 2004, the Lab opened the Isotope Production Facility (IPF), complete with a dedicated proton beam split off from the main accelerator. The IPF was part of a bigger revamp and rebrand of the Los Alamos Meson Physics Facility into today’s Los Alamos Neutron Science Center. In 2005, the IPF shipped its first commercial-scale batch of Sr-82 to a major healthcare company, proving that the new facility could not only make the vital isotope, but could deliver it at meaningful scale. Rb-82 PET imaging is more accurate and less invasive than alternative methods of diagnosing coronary artery disease, and it’s use continues to rise, with hundreds of thousands of scans done each year in the United States.
“The history of isotope production at Los Alamos is impressive, as is its national and global impact,” says Eva Birnbaum, former manager of the Isotope Production and Distribution Program at Los Alamos. “The program also made significant scientific contributions in nuclear data and radiochemical separations related to isotope production. The Isotope Production Facility itself is a flagship facility for proton-based isotope production.”
The IPF’s scope has expanded to include other isotopes for diagnostic imaging as well as isotopes to actually treat disease, like actinium-225 (Ac-225). Ac-225 is an alpha-emitter that can be targeted, via antibody or molecular tag, at tumor cells, concentrating in and destroying them with pinpoint precision. One atom of Ac-225 emits four separate alpha particles as it decays into stable bismuth-209. The alpha particles’ high energy is destructive to living cells, but their slow speed limits their range to nearby tumor cells while sparing surrounding healthy tissue. Between 2014 and 2018, Los Alamos joined Oak Ridge and Brookhaven national laboratories in a tri-lab initiative to produce Ac-225. The collaboration demonstrated the ability to close a critical supply gap that had previously left the U.S. dependent on foreign or legacy stockpiles.
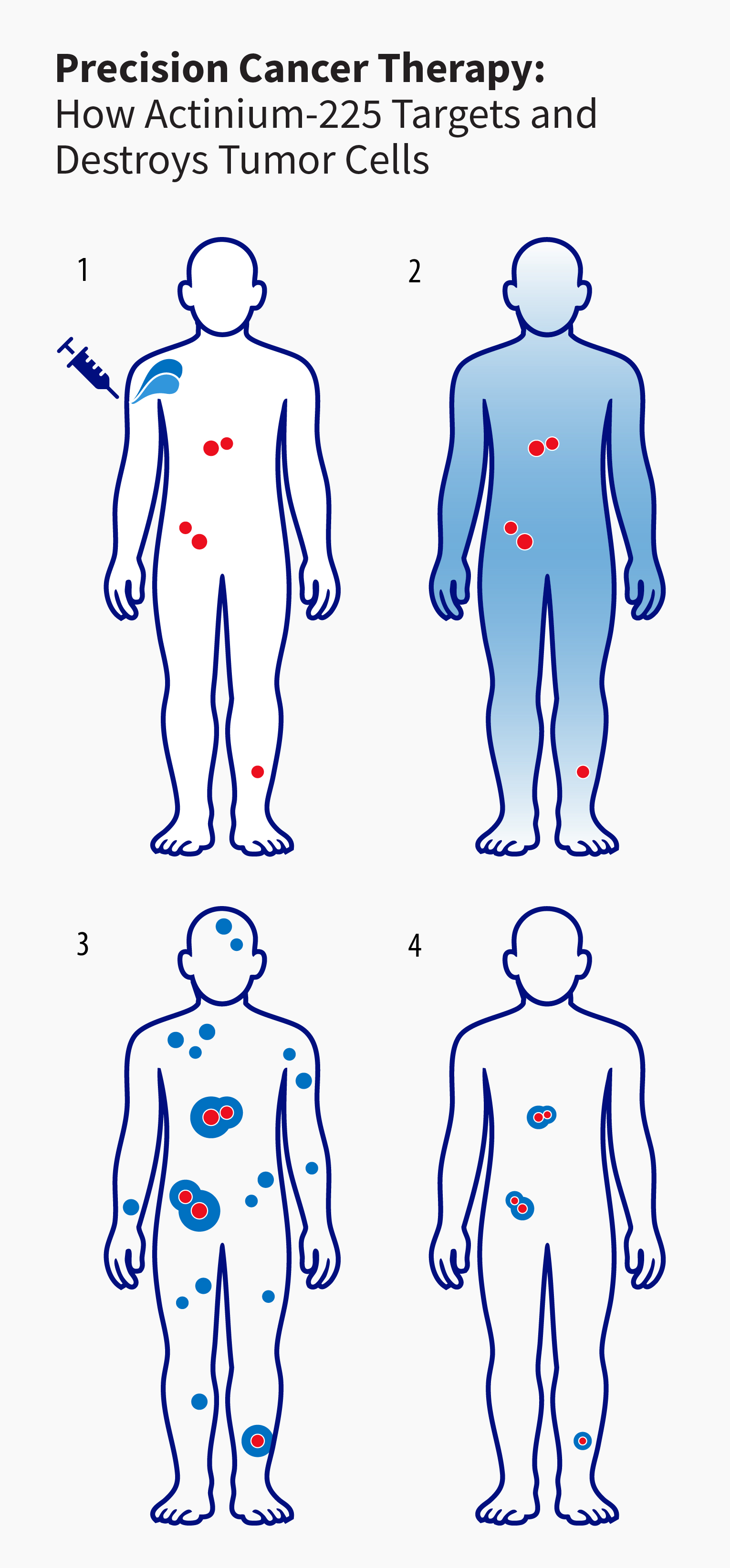
(1) The patient receives an injection of Ac-225 that has been chemically linked to antibodies or molecules that seek out tumor-specific markers.
(2) The Ac-225 circulates through the body and concentrates on tumor cells bearing the right markers.
(3) As the Ac-225 decays inside or near the cancer tissue, its alpha emissions cause lethal damage to cancer cells while sparing most surrounding healthy tissue.
(4) Over time the Ac-225 decays, eliminating the radiation source from the patient’s body and leaving tumors significantly smaller in size.
Secure supply chain
“The Lab’s accelerator-based isotope supply chain supports research institutions and commercial partners across the U.S. and lends resilience against foreign dependence or global shortages,” says Kevin John, Office of Science Program Director and also a former manager of the Isotope Production and Distribution Program at Los Alamos.
Indeed, production of Sr-82 and other isotopes has now successfully been transferred to industry, and commercial Ac-225 production is being scaled up. This transfer of established technologies to the commercial sector allows innovators at the Lab to shift focus to new isotopes for fresh applications.
Beyond isotope production, Los Alamos drives discovery in radiochemical separation, target design, and accelerator operation, while also training the next generation of scientists in nuclear chemistry and health physics. These capabilities underwrite U.S. leadership in both isotope science and biomedical technology and ensure a domestic supply of the isotopes that are essential to public health and national security.
People also ask:
- What is a medical isotope? Most medical isotopes are radioisotopes, or radioactive elements, used for diagnosing or treating diseases. In imaging, the radiation emitted by the isotope acts as a tag, or tracer, to show how organs and tissues are functioning. In therapy, a radioisotope delivers radiation directly to cancerous cells to destroy them. Nonradioactive isotopes are used in medicine as tracers for metabolic and pathologic processes.








