Fifty Years of Fuel Cell Innovation
The Los Alamos fuel cell program has contributed to advancing the industry since the 1970s.
- Rebecca McDonald, Science Writer

Hydrogen fuel cells are not new. If you remember watching the movie Apollo 13, you might recall that when an oxygen tank ruptured, it imperiled the fuel cells that gave the astronauts electricity and drinking water. Although the earliest attempts at fuel cells were made in the 1800s, space program scientists in the 1950s, along with General Motors employees, developed electrochemical fuel cells to convert hydrogen and oxygen into electrical power and water for spacecraft. These fuel cells were critical power sources on the Gemini and Apollo missions; they weighed less than batteries and could last longer. Fuel cells were key to manned missions and remained invaluable components in space travel for decades.
The Los Alamos fuel cell program made seminal innovations that underlie all of today’s transportation fuel cells
And then, during the energy crisis of the 1970s, scientists began to wonder if hydrogen fuel cells could be used for vehicles here on Earth. The spacecraft fuel cells were far too expensive to consider and did not have the durability needed for cars, but perhaps science could help. In 1977, a group of researchers at Los Alamos Scientific Laboratory (now Los Alamos National Laboratory) got a twenty-five-thousand-dollar-Laboratory Directed Research and Development grant to develop fuel cells for ground transportation. This investment launched the Lab’s fuel cell program, which proceeded to make seminal innovations that underlie all of today’s transportation fuel cells and more.
Pull out the platinum
In the beginning of the Los Alamos fuel cell program, scientists made demonstration vehicles to show off their ideas. First, they modified a passenger car and a pickup truck to use hydrogen for fuel. Then, they developed a golf cart that drove using a hydrogen-oxygen phosphoric acid fuel cell instead of a battery—it was efficient and gave off only water vapor as emissions, what could be better?

Hydrogen fuel cells are similar to batteries: they have two electrodes (anode and cathode) and use the energy from chemical bonds to drive electrons through an external circuit. Key to the technology is a polymer electrolyte membrane, which separates the electrodes and contains a catalyst to facilitate the reaction—as such, the resulting product is often referred to as a polymer electrolyte fuel cell (PEFC). (Another common term, PEMFC, refers to the polymer electrolyte membrane fuel cell or proton exchange membrane fuel cell). One of the challenges that faced Lab scientists was that the best catalyst was also the most expensive: pure platinum metal. The researchers set out to find a way to reduce the amount of platinum needed (or find an alternative catalyst), increase efficiency, and ensure the durability of the fuel cells.
In the 1980s, the Los Alamos fuel cell program grew significantly and began to make pivotal advances. One of the most important breakthroughs was the development of a thin-film, low-platinum electrode.
“This innovation dramatically lowered the required amount of platinum by a factor of more than 20, while simultaneously improving performance,” says Rod Borup, recently retired Los Alamos fuel cell scientist and Laboratory Fellow. The 1980s team also improved the structure and composition of the thin-films to ensure their durability without loss of performance. Fuel cell manufacturers worldwide currently use this low-platinum approach.
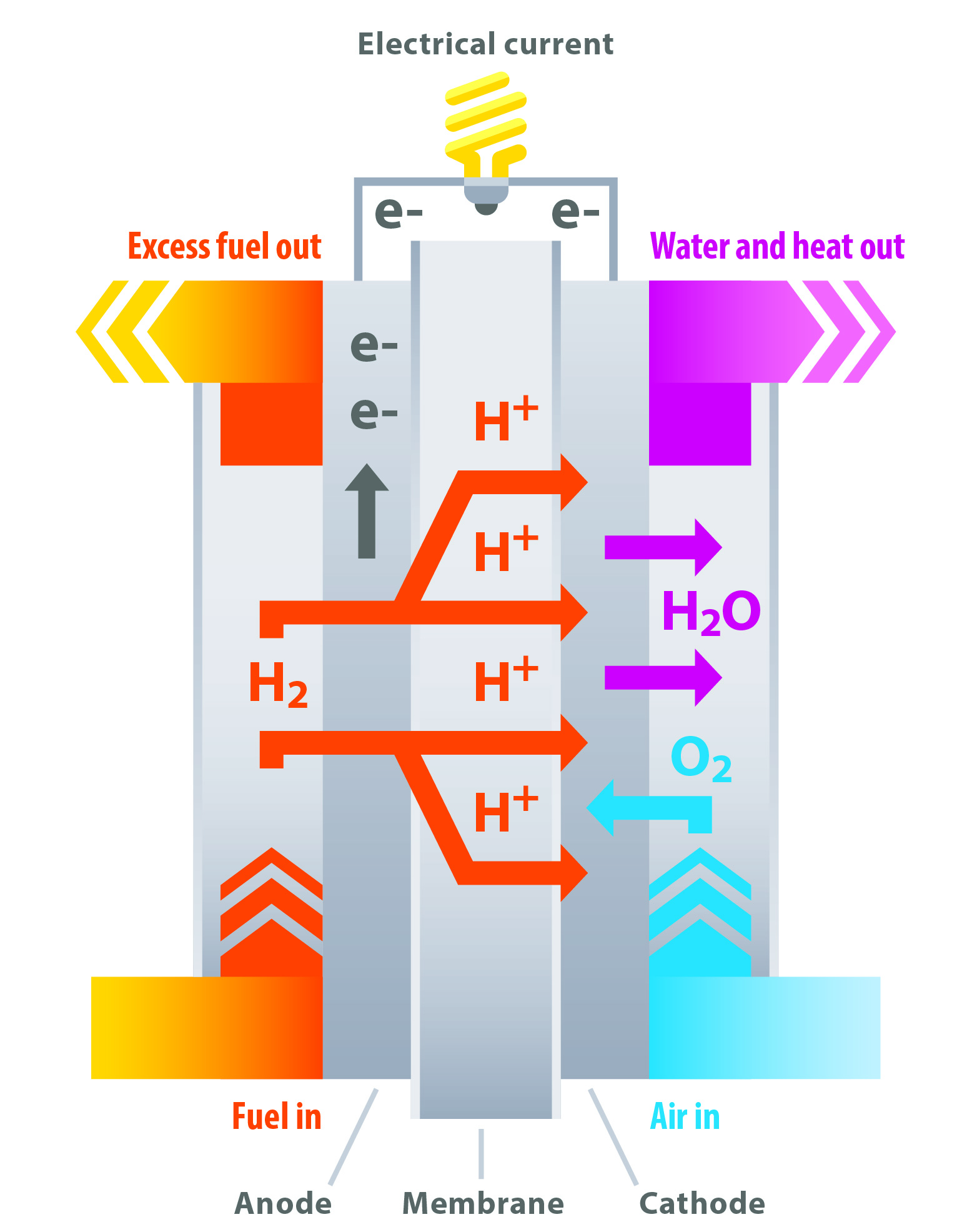
Hope for hydrogen
In 1991, Los Alamos established the General Motors Joint Development Center. This collaboration was an effort funded by General Motors and the Department of Energy (DOE) to facilitate the development of an electrochemical engine—a PEFC system fueled by hydrogen-rich gas produced on demand from a methanol precursor. This approach stood to mitigate the challenge of hydrogen availability, as the country lacked infrastructure to provide on-demand purified hydrogen.
However, the process of converting methanol to hydrogen-rich gas sometimes left carbon monoxide in the mix. This impurity in the hydrogen negatively impacted fuel cell performance. Los Alamos scientists mitigated this challenge by developing PEFCs that could operate using impure hydrogen fuel. The scientists figured out that by bleeding low levels of air into the fuel feed stream, they could remove the carbon monoxide catalytically. This development paved the way to more widespread use of PEFCs.
Over the decades, the Los Alamos fuel cell team continued to collaborate with industry partners to further improve fuel cells, looking to develop new platinum-free cathode catalysts, to improve fuel cell lifetime and durability, and also to innovate around new sources of hydrogen. In 2016, Los Alamos partnered with Argonne National Laboratory to form the ElectroCat Consortium dedicated to investigating non-platinum-group metal catalysts.
“Even with our thin-film electrode approach, the platinum needed still accounts for about 40 percent of the cost of the fuel cell,” says Los Alamos electrochemist and Lab Fellow Piotr Zelenay, who continues to lead the Consortium. “Replacing it with a platinum-group-metal-free catalyst would give the ultimate savings.”
Beyond cars
Around 2014, some scientists began to shift focus to the nearly four million diesel semitrucks driving back and forth across America.
“The DOE fuel cell program switched to heavy duty vehicles and trucks,” says Zelenay. “For trucks, batteries are not practical due to their prohibitive size and weight, but hydrogen fuel cells showed potential as an alternative solution.” In 2020, the DOE launched a consortium, co-led by Los Alamos and Lawrence Berkeley national laboratories, to advance new types of fuel cells.
Scientists faced new challenges to adapt hydrogen fuel cells for heavy-duty vehicles. The fuel cells used in passenger cars operate at a specific temperature, but the amount of power needed to drive a semitruck would produce excess heat and render the fuel cell inoperable. Developing high-temperature hydrogen fuel cells would require implementing new types of materials, which they did.
Among the growing list of achievements, one group of Los Alamos researchers produced and patented a completely new type of membrane and electrode combination called “protonated PWN70,” which stands to accelerate the use of fuel cells for heavy-duty vehicles. The new materials are not only efficient at the right temperatures but also durable. Today, the consortium, which is called Multi Modal Fuel Cell Technologies (M2FCT), continues to focus on future energy needs. Although it is no longer prioritizing trucks, the scientists are looking broadly at energy needs and even stationary applications.

“Fuel cells are inherently scalable,” says Borup. “Through our decades of experience working on polymer electrolyte fuel cells, we can adapt these technologies to anything from portable electronics to homes.” In fact, they’ve already demonstrated it. In the early 2000s, Los Alamos fuel cell scientists worked with a few industry partners to improve performance and miniaturize direct methanol fuel cells (DMFCs), which rely on high-density liquid methanol and don’t require conversion to hydrogen. Small, portable DMFCs are attractive as lightweight alternatives to batteries for laptop computers and other small electronics.
Fuel cells are inherently scalable and could be adapted to anything from portable electronics to homes
The fuel cell story is far from complete. In 2014, Toyota began to market its hydrogen fuel cell car, the Mirai (“future” in Japanese), in Japan and California. Today, thousands of fuel cell–powered cars are being used in the U.S., and a handful of car manufacturers have fuel cell vehicles for sale in various markets around the world.
For nearly five decades, Los Alamos scientists have demonstrated success in developing hydrogen fuel cells for any need, and in the decades to come, one can only imagine the innovative ways that fuel cells will be a part of our lives.

People Also Ask:
- What is a fuel cell? A fuel cell is a device that generates electricity using the chemical energy from various types of fuel instead of burning the fuel.
- How does a hydrogen fuel cell work? A hydrogen fuel cell combines hydrogen gas and oxygen in an electrochemical reaction to produce electricity, water, and heat.








