Advancing Understanding of Actinides in Aqueous Media
- Zach Jones

Advancing our understanding of actinides in aqueous media could help solve many outstanding problems in actinide science. Almost all aspects of technologically relevant actinide chemistry rely—at some point—on aqueous actinide processing. The impact spans from large-scale plutonium metal production to actinide environmental monitoring and efforts to achieve energy security using nuclear power. Unfortunately, we have a real knowledge gap in this area—in particular, speciation and reactivity of actinides in industrially relevant aqueous solutions is often poorly defined. One example is actinide species dissolved in buffered ammonium acetate-acetic acid solutions—beyond a few focused studies, this commonly used stock solution is poorly characterized because of the inherent challenges associated with handling highly radioactive actinides. Interpreting spectroscopic results from actinides in this complicated aqueous environment is also challenging compared to well-defined organic solutions and solid-state crystals. Nevertheless, the absence of knowledge in this area is surprising given that actinide-containing ammonium acetate-acetic acid solutions facilitate small-scale production of actinide metals, serve as starting materials for labeling chelators with alpha-emitting radionuclides for therapeutic applications, and are used in both aqueous synthesis and, to a more limited extent, actinide separations.
To explore actinide speciation in buffered ammonium acetate-acetic acid (pH = 5.5) stock solution starting materials, we chose environments where the solvent mixture was present at three different concentrations. Our intent was to obtain results that were directly relevant (or easily extrapolated) to current uses in the laboratory. These three different concentrations were dilute (0.1 M), intermediate (4 M), and concentrated (15 M). In the dilute solution, the actinide and acetate ions were solutes in an aqueous matrix, while in the intermediate solution, there was an abundance of coordinating acetate ions in an excess of water. In the concentrated solutions, however, water was of sufficiently low concentration that it was a solute rather than a solvent. The primary analytical method that we used was solution-phase X-ray absorption spectroscopy (XAS), and we evaluated the results using structural data from single crystals grown from the acetate solutions. This included characterization of M(acetate) (M = americium, curium, and europium; the latter is a nonradioactive surrogate for the exploratory studies) using single-crystal X-ray diffraction.
For the X-ray absorption spectroscopy experiments, we examined two actinides on either end of the scale in the actinide series, in which ions get progressively smaller with increasing atomic number (known as the actinide contraction). Specifically, we chose the largest trivalent 5 element, actinium (Ac, ionic radius of 1.12 Å in six-coordinate complexes), and, in the middle of the series, curium (Cm, ionic radius of 0.97 Å in six-coordinate complexes), one of the smallest actinides that can be reasonably obtained in milligram quantities. By choosing these elements, we could examine the effect of actinide cation size, and by extension Lewis acidity, on coordination chemistry in these solutions.
Crystal structures
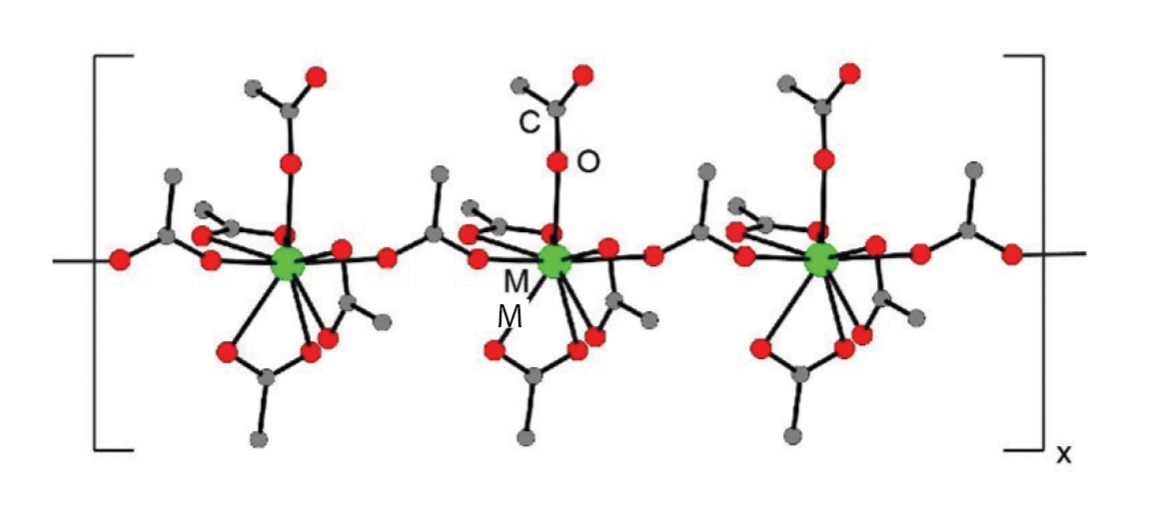
Solid-state structures of Am(acetate), Cm(acetate), and Eu(acetate) were isolated from slow evaporation of the trivalent metal cations dissolved in a 10 M aqueous solution of ammonium acetate. The M(acetate) compounds were isomorphous (crystallized in the same form; fig. 1) with three bidentate, terminal acetate ligands (coordinated in two positions), one monodentate and terminal (bound in one location), and one monodentate and bridging. This gives an overall nine-coordinate geometry with no bound water molecules.
X-ray absorption spectroscopy
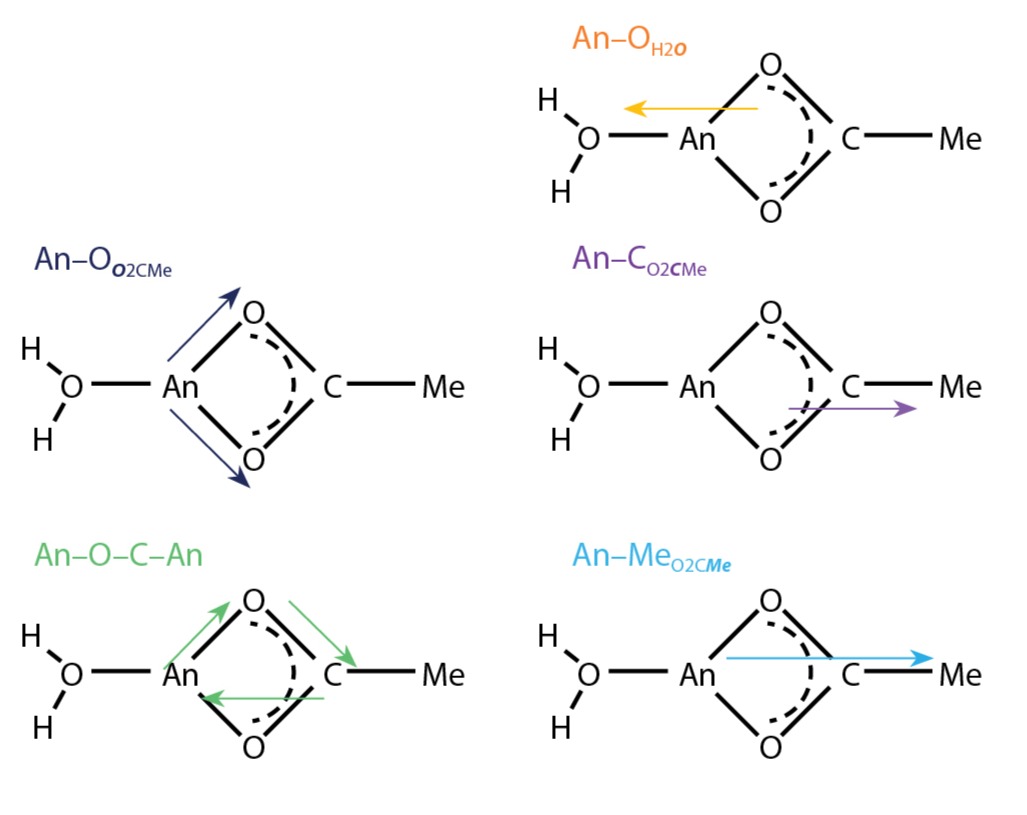
To determine if the solid-state structures were maintained in solution, we used the complimentary techniques of L-edge X-ray absorption near edge spectroscopy (XANES) and L-edge extended X-ray absorption fine structure (EXAFS) spectroscopy. X-ray absorption spectroscopy is well suited to characterize actinides in solution because it is atom-specific and provides information on oxidation state(s) and local structure even with very low concentrations of metal analytes (micrograms of actinium and milligrams of americium and curium). In particular, while XANES is very sensitive to the oxidation state and coordination chemistry of the absorbing atom, EXAFS is useful for determining distances, coordination number, and species of the neighbors of the absorbing atom. Fig. 2 displays a cartoon representation of the interactions that occur between the analyte and its neighboring atoms. All of the X-ray absorption spectroscopy measurements were performed at the Stanford Synchrotron Radiation Lightsource, beamline 11-2, which is optimized for challenging X-ray measurements such as those with dilute or radioactive samples.
XANES results
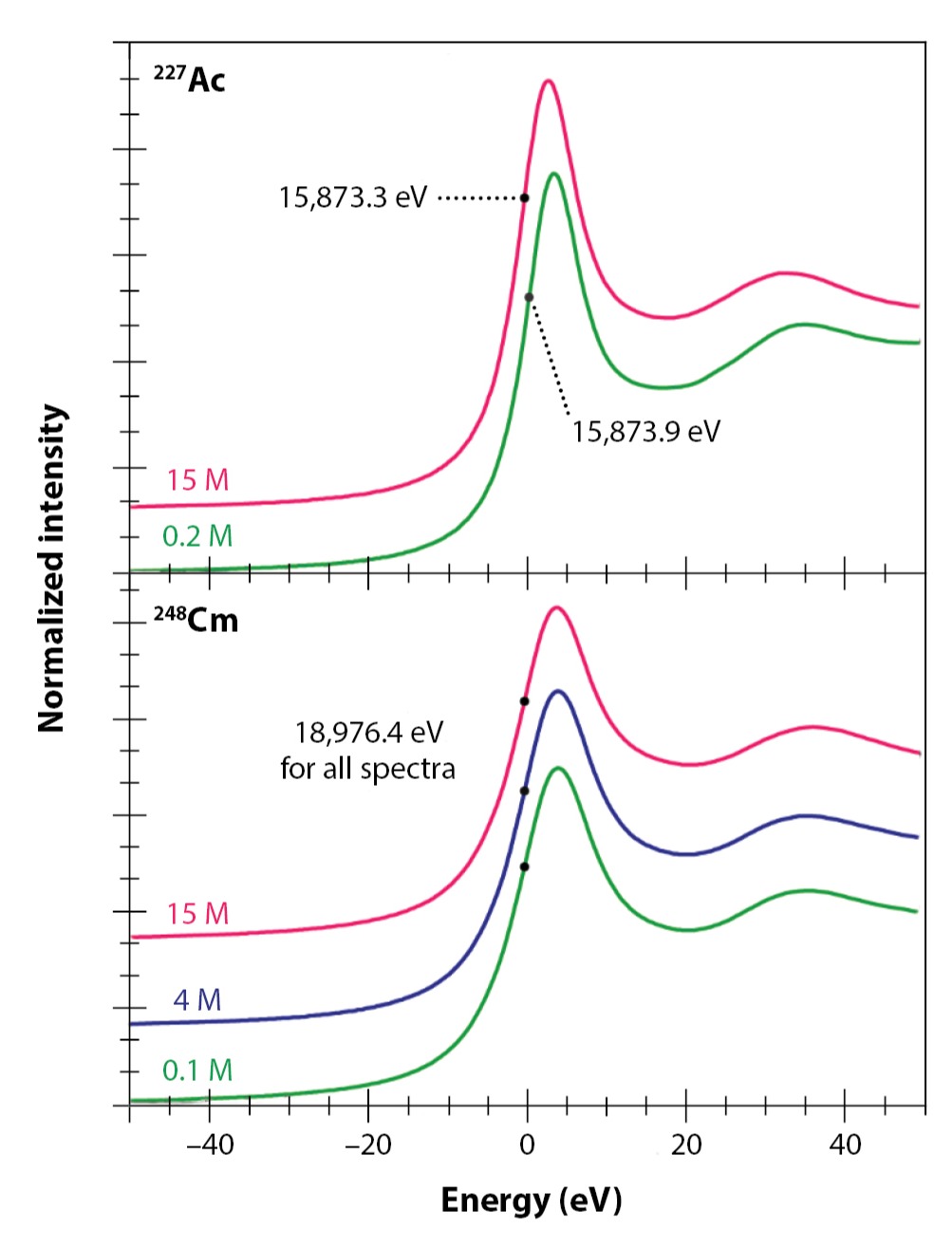
The L-edge XANES spectra in all three acetate solutions (dilute, intermediate, and concentrated) were dominated by an absorption peak superimposed on an absorption edge for both elements (Fig. 3). The inflection point for actinium(III) in dilute acetate solution was higher than the value recorded in concentrated solution (15,873.9 versus 15,873.3 eV) and an even greater difference was observed between it and the previously reported aquo ion (15,874.3 eV). We did not see this for curium. Instead, the curium(III) inflection points from all three acetate solutions were identical to the value previously determined for the aqua ion in dilute nitric acid. It is unclear why the actinium inflection points are more sensitive to ligand environments than curium, although one possible explanation is that they derive from more substantial orbital mixing in the actinium-ligand bonds (see for a description of the relationship of actinide atomic number and interatomic bonding).
XANES alone did not offer substantial insights, as it is only sensitive to the oxidation state and electronic structure of the absorbing atom. Therefore, we turned to EXAFS to help determine the structure around the absorbing atom.
EXAFS results
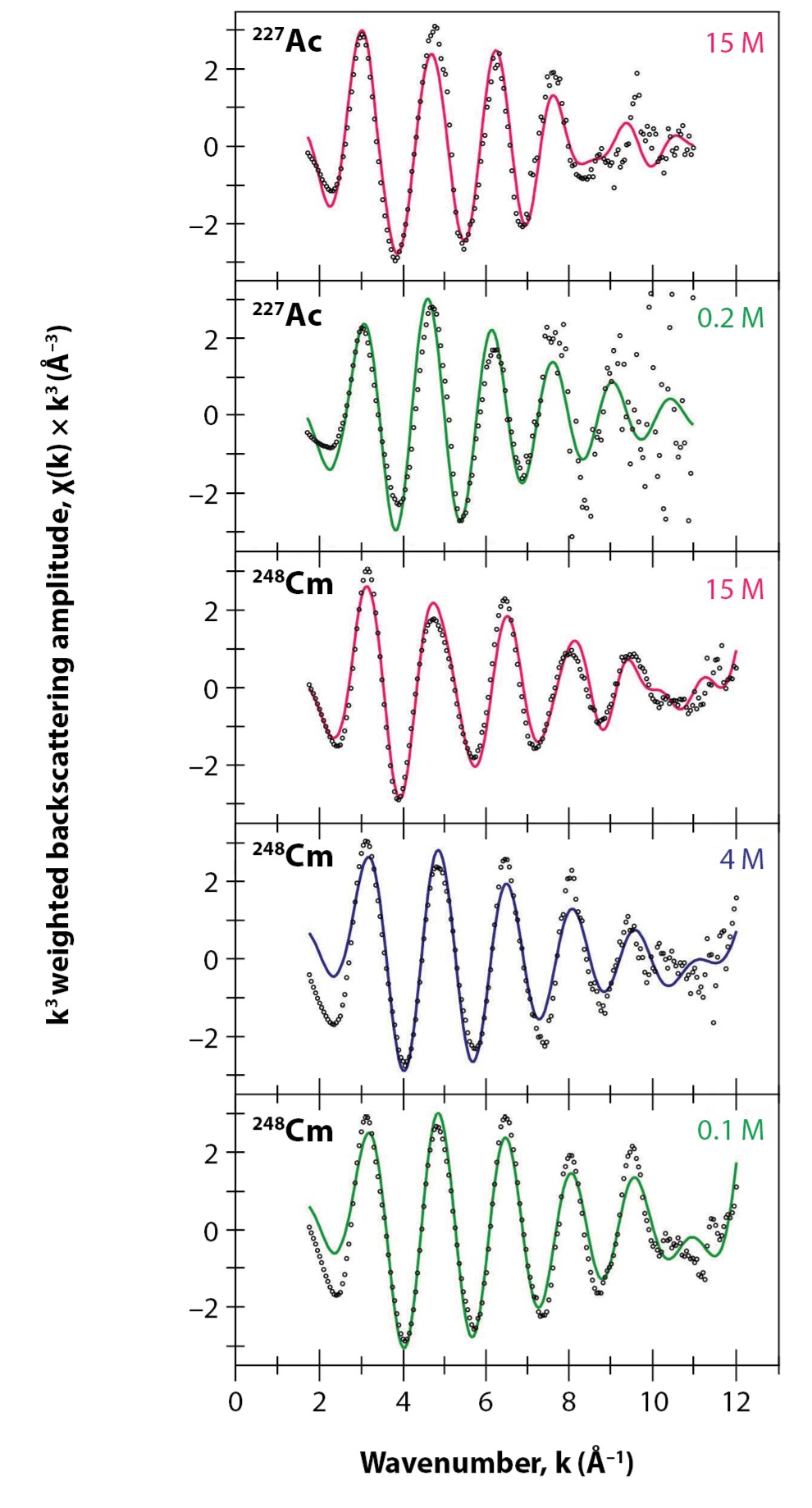
For dilute acetate solutions, structural metrics from the actinium(III) L-edge EXAFS data spectra were consistent with Ac(HO), equivalent to the two other literature reports and in agreement with predictions from molecular dynamics simulations. We also obtained a straightforward result for curium(III), as the data indicated eight coordinated water molecules, comparable to previous reports on the curium(III) aquo ion.
Concentrated solutions were, however, distinct from dilute, giving EXAFS spectra that were inconsistent with a single-ligand environment. Instead, multiple contributions associated with actinide-bound acetate were present (Fig. 4). For actinium, there were two well-resolved and short Ac–O scattering pathways in the innermost coordination sphere, corresponding to both water and acetate ligands. After carefully modeling the data, we found an average stoichiometry of six water molecules and three acetate ligands, i.e., Ac(HO)(acetate).
For curium, the low-frequency contributions refined as a single environment of nine oxygen atoms. Longer-range electron scattering was modeled with 4.5 acetate ligands (to total nine oxygen donor atoms), giving Cm–O bond distances that were consistent with the X-ray diffraction data for nine-coordinate Cm(acetate). Overall, these data suggest that the curium(III) inner coordination sphere is dominated by acetate in concentrated solutions, with minimal contributions from bound HO.
Small changes in Lewis acidity impact actinide coordination chemistry
Our acetate studies align with the common assumption (and a limited number of experimental observations) that actinide(III) complexation is directly influenced by Lewis acidity (i.e., the strength of their attraction to electron donors). Curium, a stronger Lewis acid, showed an inner coordination sphere populated by approximately four acetate ligands in concentrated solutions where acetate ions were in high abundance. For the larger actinium cation, a weaker Lewis acid, the structure contained only three acetate ligands, formulated as neutral Ac(H2O)6(acetate)3.
This provides rare experimental data showing how 5-element speciation varies as actinide ionic radii contract, analogous to behavior observed for rare earth elements. Although this is expected, the impact of the actinide contraction on coordination chemistry is scarce in the literature, likely due to the difficulties of handling these highly radioactive elements and performing detailed solution-state measurements. The implications of speciation differences in these acetate solutions are subtle but important. They highlight that the dominant species present in high-concentration solutions for the early actinides is not equivalent to that present for the minor actinides. We speculate that the increased complexation tendencies for the late actinide(III) cations likely persists even when the ammonium acetate-acetic acid concentration is less than 15 M. It seems possible that acetate prefers binding small -elements (e.g., curium(III)) over large actinides (e.g., actinium(III)) between 4 and 15 M solution concentrations.
Summary
A range of X-ray absorption spectra were collected for actinium(III) and curium(III) in aqueous, buffered solutions of ammonium acetate-acetic acid at three different concentrations relevant to current laboratory applications. At low concentrations, the structures were found as the known aquo ions, Ac(H2O)93+ and Cm(H2O)83+. At high concentrations, where acetate ions are abundant, the actinium species was characterized as neutral Ac(H2O)6(acetate)3, whereas the curium solution showed an inner coordination sphere populated by approximately four acetate ligands. This increased affinity is likely due to the higher Lewis acidity of the curium(III) ion, which has a smaller ionic radius.
Better characterization of these aspects of aqueous actinide coordination chemistry would provide researchers with critical information about how active species in solution change with conditions, potentially having significant impact. We hope that this work motivates further study of actinides in relevant aqueous solutions.
Zach Jones
Originally from Portland, Oregon, Zach received a BS in chemistry while conducting undergraduate research on inorganic solid-state superconducting materials with Prof. David Johnson at the University of Oregon. Zach then attended the University of California, Santa Barbara for his PhD work, probing structure-property relationships of nanoscale heterogeneous catalysts with Prof. Susannah Scott. During one of his many synchrotron trips at the Stanford Synchrotron Radiation Lightsource, Zach met Stosh Kozimor, which led to the postdoctoral position and research described herein. Zach is now a Scientist in Sigma division with Fabrication Manufacturing Science (Sigma-1).
Acknowledgments
Many thanks to Maryline G. Ferrier, Benjamin W. Stein, Laura M. Lilley, Stosh A. Kozimor, Maksim Y. Livshits, David H. Woen, Elodie Dalodière, Veronika Mocko, Frankie D. White, Karah E. Knope, Brian L. Scott, and Jennifer N. Wacker for their contributions. These contributions included project conceptualization, writing, editing, acquisition of XAS data, single crystal structure refinement, actinide purification, and optical measurements. We also thank the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Heavy Element Chemistry program (2020LANLE372) and LANL's LDRD-DR (20180005DR and 20190364ER). Postdoctoral support was provided by the Glenn T. Seaborg Institute. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, was supported by the U.S. DOE, Office of Science, Office of Basic Energy Sciences under Contract No. DE-AC02-76SF00515.
Further reading:
- Z.R. Jones, M.Y. Livshits, F.D. White, E. Dalodiere, M.G. Ferrier, L. M. Lilley, K.E. Knope, S.A. Kozimor, V. Mocko, B.L. Scott, B.W. Stein, J.N. Wacker, F.D. White, “Advancing understanding of actinide (III) (Ac, Am, Cm) coordination chemistry in ammonium acetate and acetic acid buffered aqueous stock solutions,” Chem. Sci., 2021, 12, 5638.
- M.G. Ferrier, B.W. Stein, S.E. Bone, S.K. Cary, A.S. Ditter, S.A. Kozimor, J.S. Lezama Pacheco, V. Mocko, G.T. Seidler, “The coordination chemistry of CmIII, AmIII, and AcIII in nitrate solutions: an actinide L3-edge EXAFS study,” Chem. Sci., 2018, 9, 7078.
- M.G. Ferrier, B.W. Stein, E.R. Batista, J.M. Berg, E.R. Birnbaum, J.W. Engle, K.D. John, S.A. Kozimor, J.S. Lezama Pacheco, L.N. Redman, “Synthesis and characterization of the actinium aquo ion,” ACS Cent. Sci., 2017, 3, 176.








